A DUE TRIAL
A DUE Is the First and Only
Phase 3 Clinical Trial of a
Single-Tablet Combination Therapy vs ERA or PDE5
Inhibitor Monotherapy for PAH WHO FC II-III1
Study design: Multinational, multicenter, double-blind, randomized, and active-controlled phase 3 study of a once-daily, single-tablet dual combination of macitentan 10 mg and tadalafil 40 mg. The primary endpoint was change from baseline in pulmonary vascular resistance at Week 16.2
(OPSYNVI® [n=70] vs macitentan
[n=35])2
REDUCTION
Primary Endpoint | Ratio of Geometric Means 0.71; P<0.0001 (95% CL; 0.61-0.82)
(OPSYNVI® [n=86] vs tadalafil
[n=44])2
REDUCTION
Primary Endpoint | Ratio of Geometric Means 0.72; P<0.0001 (95% CL; 0.64-0.80)
Treatment with OPSYNVI® (macitentan/tadalafil) resulted in greater reductions in PVR compared with macitentan and tadalafil alone2
STUDY DESIGN
Patients were randomized into treatment arms depending on their treatment status at baseline1


Select inclusion criteria1:
- mPAP ≥25 mmHg
- PAWP or left ventricle end-diastolic pressure ≤15 mmHg
- PVR ≥3 Woods units (ie, ≥240 dyn•s/cm5)
- WHO FC II-III
- PAH-specific treatment-naïve or on a stable dose of ERA or PDE5 inhibitor monotherapy for at least 3 months
BASELINE CHARACTERISTICS
Patient demographics at baseline (N=187)1,2
Treatment History
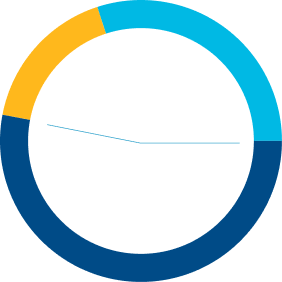

47%
Previously
treated
53%
Treatment
naïve
Etiology
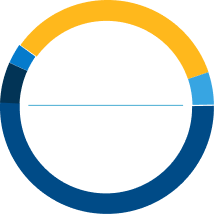

35%
PAH-CTD
51%
Idiopathic
PAH
PAH
Mean Age
50
years (range 18-80)
Gender
78%
Female
22%
Male
WHO Functional Class
51%
FC II
49%
FC III
A DUE TITRATION
Titration used in A DUE trial1
The double-blind treatment period began with a 2-week tadalafil titration phase followed by a maintenance phase:
- Patients received macitentan 10 mg once daily, tadalafil 20 mg once daily, or both as separate tablets, plus respective placebos
- Patients who were receiving a stable dose of a PDE5 inhibitor at baseline did not require titration, and tadalafil 40 mg was administered
- Tadalafil was up-titrated to 40 mg once daily
Phase
- Started on Day 15
- Patients received either OPSYNVI®, macitentan 10 mg once daily, or tadalafil 40 mg once daily, along with respective placebos depending on treatment arm
Prespecified stable doses were1:
40 mg
tadalafil60 mg to
120 mg
sildenafil10 mg
vardenafilReview safety
and see how OPSYNVI® compares to monotherapy