Dosage and Administration
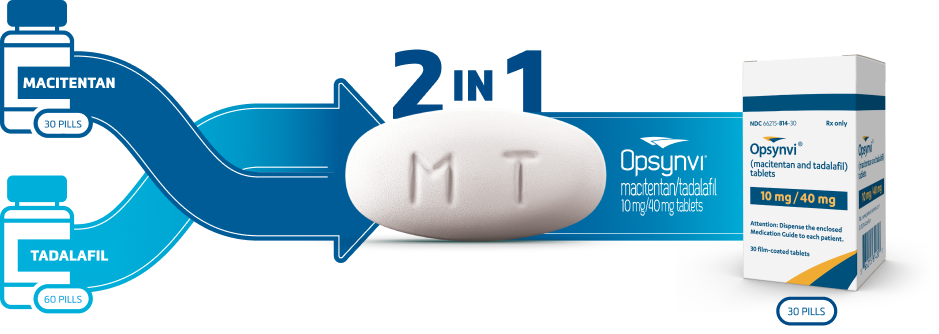
OPSYNVI® Contains 2 Proven Therapies in 1 Tablet1,2
OPSYNVI® reduces pill count and may help reduce costs compared with separate ERA and PDE5i prescriptions1-7
With a once-daily tablet, OPSYNVI® (macitentan/tadalafil) delivers guideline-recommended macitentan and tadalafil.1,2
How many tablets do you think patients may take in a single week with separate ERA and PDE5i prescriptions?
Combination therapy with a single prescription
OPSYNVI® is the first and only US FDA-approved single-tablet combination of 2 recommended treatments for PAH1,8

- For females of reproductive potential, exclude pregnancy prior to initiation of therapy and ensure use of effective contraceptive methods prior to the initiation of treatment, during treatment, and for 1 month after discontinuation1

1 once-daily tablet
- OPSYNVI® can be administered with or without food1
- Tablets should not be cut, crushed, or chewed1
- If the patient misses a dose of OPSYNVI®, tell the patient to take it as soon as possible and then take the next dose at the regularly scheduled time. Tell the patient not to take 2 doses at the same time if a dose has been missed1
- The use of OPSYNVI® is not recommended in patients undergoing dialysis. Avoid use of OPSYNVI® in patients with severe renal impairment1
- OPSYNVI® was not studied in patients with severe hepatic impairment. OPSYNVI® must not be initiated in patients with severe hepatic cirrhosis (Child-Pugh class C) or clinically significant elevated hepatic aminotransferases (>3 x ULN)1
| Medication1 | Effect1 |
|---|---|
| Nitrates |
|
| Strong inducers of CYP3A4 (eg, rifampin) |
|
| Strong CYP3A4 inhibitors (eg, ritonavir, ketoconazole, and itraconazole) |
|
| Moderate dual or combined inhibitors of CYP3A4 and CYP2C9 (eg, fluconazole and amiodarone) |
|
| Alpha-blockers |
|
| Antihypertensives |
|
| Alcohol |
|
Explore the A DUE trial
and how OPSYNVI® offered greater reductions in PVR vs monotherapy1
